Global Health Work Continues with New Grant Awards
https://www.thearcadygroup.com/wp-content/themes/crocal/images/empty/thumbnail.jpg
150
150
The Arcady Group
https://www.thearcadygroup.com/wp-content/themes/crocal/images/empty/thumbnail.jpg
Smart Medication Container Included in GDF Product Catalog
https://www.thearcadygroup.com/wp-content/uploads/2020/11/Screen-Shot-2020-11-12-at-12.40.12-PM.png
565
362
The Arcady Group
https://www.thearcadygroup.com/wp-content/uploads/2020/11/Screen-Shot-2020-11-12-at-12.40.12-PM.png
M4ALL Selected as Key Partner for Major BARDA Grant
https://www.thearcadygroup.com/wp-content/uploads/2020/05/Copy-of-Untitled-3.png
940
788
The Arcady Group
https://www.thearcadygroup.com/wp-content/uploads/2020/05/Copy-of-Untitled-3.png
Publication on China’s Use of MERM Technology To Support Differentiated Care
https://www.thearcadygroup.com/wp-content/uploads/2020/05/MERM-3.jpeg
1000
899
The Arcady Group
https://www.thearcadygroup.com/wp-content/uploads/2020/05/MERM-3.jpeg
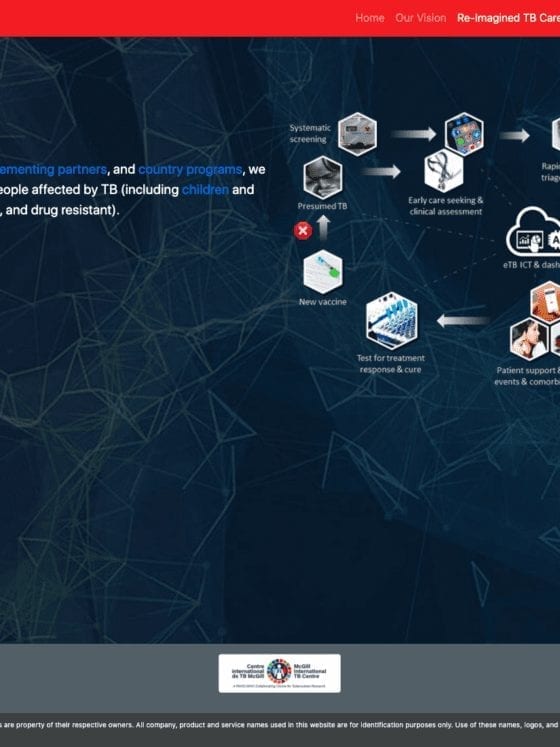
Stop TB Partnership Launches Virtual Innovation Spotlights
https://www.thearcadygroup.com/wp-content/uploads/2020/04/STBP_ReimaginingTBCare_Qure_Apr20-pdf.jpg
1497
1058
The Arcady Group
https://www.thearcadygroup.com/wp-content/uploads/2020/04/STBP_ReimaginingTBCare_Qure_Apr20-pdf.jpg
Arcady Group Partners With Stop TB Partnership and McGill University to Launch Re-Imagining TB Care Website
https://www.thearcadygroup.com/wp-content/uploads/2020/03/TBCare_Website-2.jpg
2016
1101
The Arcady Group
https://www.thearcadygroup.com/wp-content/uploads/2020/03/TBCare_Website-2.jpg
Arcady Group’s Founder Selected As Team Lead For India’s Integrated Digital Adherence Technology Evaluation
https://www.thearcadygroup.com/wp-content/uploads/2019/11/Mewat-Team-1-1024x576-1024x576-1.jpg
1024
576
The Arcady Group
https://www.thearcadygroup.com/wp-content/uploads/2019/11/Mewat-Team-1-1024x576-1024x576-1.jpg